I’m writing this as we approach autumn in 2022, with general increases in the cost of living as well as the spectacularly high increases in energy costs projected for the UK from October (and then again in January). Yes, this is a cost of living crisis. And yes, energy is but one – fairly large – part of this. Mindful of the fact that many will not be able to afford even small increases in the cost of energy, nevermind what is predicted for October, I’m going to describe several approaches to minimising energy that can be applied as possible.
The first consideration is to identify where your main sources of energy usage are. Typically it would include:
- Central heating (gas or electric)
- Heating water
- Cooking
- Freezers and refrigerators
- Washing machine
- TV
- Dishwasher (if you have one)
- Tumbledryer (if you have one)
- Computers (if you have/use one at home)
- Other small electronics (phones, tablets)
Let’s start with central heating.
a) If you have a condensing combi boiler without a hot water cylinder: by turning down the flow temperature on your boiler you could save between 6-8% on your gas usage. The flow temperature is the temperature of the water passed out to the radiators (not set on the thermostat) and the temperature at which it returns. By lowering the flow temperature you can set the system to run in ‘condensing’ mode more than it may currently be doing, which increases the overall efficiency of the system. Usually it runs at 80/60: 80 degrees Celsius out to the radiators and 60 degrees Celsius on return.
However this is tricky to get right, and would usually be set by an engineer during a service (so worth asking about). If you do reduce the flow temperature note that it will increase the time it takes for your rooms to warm up. In other words, you’re trading speed (of heating) for efficiency.
b) Explore the use of valves on your radiator, to tailor the level of heating in each room. These come in two general types:
(1) Manual valves – which adjust the flow of water to each radiator.
(2) Thermostatic Radiator Valves (TRVs) – which control for the temperature in a specific room.
You could choose to only heat rooms that you are using, or if the thermostat in your house is in a room that is usually warmer/colder than the rest of the house, it allows you to adjust each room as needed. (For example, I usually close the valve in my office as work most of the way as I prefer it cooler than most.)
Heating water
As with your boiler, you have some choice on the temperature of hot water (that will vary depending on your system):
- With a combi, for example, the hot water is heated as you need it to a set temperature.
- With immersion tanks you heat the ‘tank’ and use this throughout the day (or until you switch it on again). These should be surrounded by thermal insulation to prevent loss of heat as you wait to use the hot water. For newer tanks this insulation is integrated into the design with solid foam; for older tanks it will be more obvious (and is worth checking that the insulation is still in good condition).
Cooking
There are several options now for cooking meals, with a wide variety of energy costs associated with them. The most efficient of these will be the microwave, air fryer and pressure cookers. Part of the reason for this is the reduced volume of space that needs to be heated (you can often feel the kitchen warm up as you cook a roast, for example). Another element is that less steam is lost to the environment during cooking (so that heat can be used to keep cooking your meal).
As a ballpark figure, an Instant Pot Pressure cooker, or air fryer uses about 10% of the energy that an electric oven would to cook the same meal.
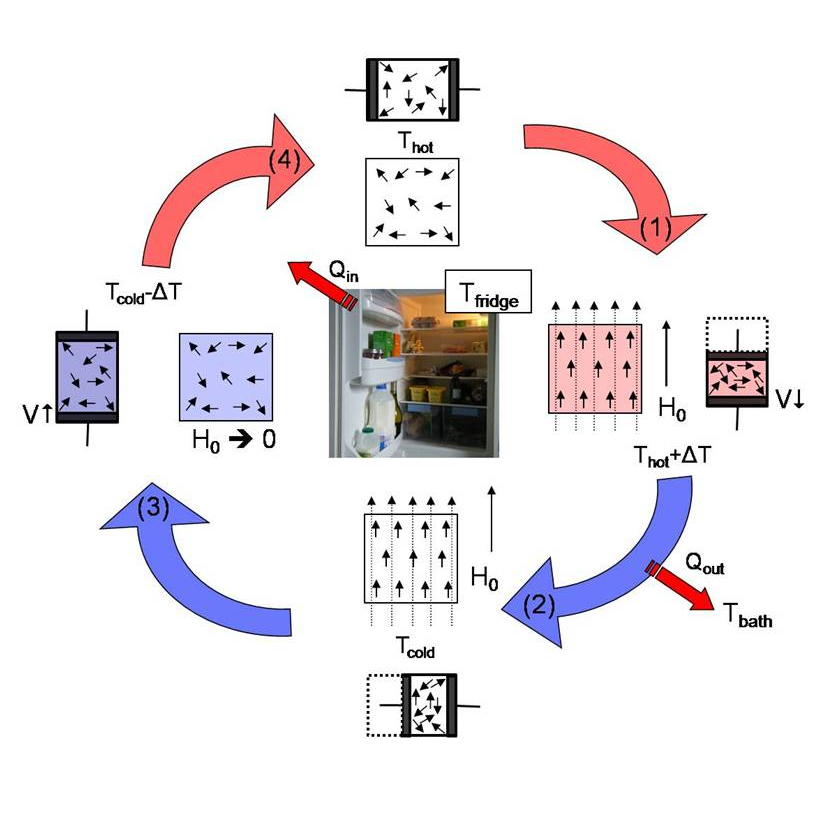
Freezers and Refrigerators
The main aim of a freezers or refrigerator is to keep a volume of space cooler than the rest of the house: the freezer is usually -18 degrees Celsius, whilst the fridge is usually +1-4 degrees Celsius. After heating (and possibly depending on your usage, cooking), your refrigeration will likely be the largest consumer of energy in your household. This stems from the difficulty in moving heat from one area to another (it’s not particularly efficient!) and will obviously be impacted on how often that fridge/freezer door is opened throughout the day as you let heat in. Whilst there have been advances in the efficiency of these appliances over the last 20 years, they can easily use 1 kWh per day (which on an electricity rate of ~50p/kWh, would equate to £182.5 per year).
To ease the burden on your fridge/freezer, pick a location in the house which is the coolest (it is easier to move heat to a cooler room than a warmer one), make sure that there is some space between the ‘fins’ at the back of the fridge/freezer and the wall (so air can flow past), and make sure that the seals on the door are kept clean. All of this should help to keep it running as efficiently as it was designed to.